CRISPR screen with an ebony marker
All of these flies are from the same parents, so why does the one on the lower right look so different? The fly on the right has a mutation in the ebony gene, which our lab is using to help screen for flies where our gene "C" may be knocked out through the gene-editing technique CRISPR.

In my previous entry we saw that RNA interference, or RNAi, of gene C resulted in a partial rescue of degeneration caused by over-expression of the Toll protein in Drosophila melanogaster eyes.
The problem with RNAi is that there is variability in the amount of gene knockdown, and so the amount of resulting protein "C" is also variable. What we want to know is, how much of an effect does protein "C" have when it is completely and totally absent, not just present in reduced levels?
To answer this question we used another molecular technique called CRISPR. Through CRISPR we are able to genetically engineer flies that
have the C gene completely deleted, rather than simply reduced.
Making a gene-specific CRISPR animal is not a short process. First I needed to clone a small piece of gene C into a special circular piece of DNA called a plasmid. To do so, we first needed DNA coding for gene C, which Professor Dolph achieved by designing two strings of oligo primers that, when hybridized together, make our gene. Then these two oligo strings--sense and antisense--are hybridized together in a step called annealing, where the two form a piece of gene C DNA.
Next, I need to put that piece of DNA into something that can be integrated into fly gametes, and we can do this using the circular plasmid DNA. Plasmid DNA can be thought of as a necklace with different beads that have different functions or outcomes when the "bead" information is transcribed during DNA transcription and translation. There are messages for starting, stopping, eye color, antibiotic resistance, and other useful directives.
To get our piece of DNA into a guide-RNA plasmid that could be read by CRISPR gene editing machinery, I used DNA scissors called restriction enzymes to cut the plasmid at specific sites. The restriction digest leaves pieces of unhybridized DNA at the cut ends. Then I can mix the cut plasmid, my gene C DNA fragment, and another enzyme called ligase in a ligation reaction that, when all the molar ratios are optimal, will cause the fragment to hybridize to the sticky ends and integrate into the plasmid as a new bead.
Once my gene was ligated into the plasmid, or DNA necklace, I then was able to transform some of my necklaces into bacteria. The bacteria then made a million necklaces for me using their replication machinery each time they divided in a bacterial culture grown overnight. The trick then is to get the necklaces out of the bacteria, and we do that using a technique called a miniprep that basically bursts open the bacteria and isolates the plasmid DNA in pure form.
Then I sent our DNA to a company in California called Best Gene, where they injected Drosophila with our DNA to integrate the DNA into the fly's genetic material. The problem is that not every fly that is injected will actually integrate the DNA. So we needed a way to screen all the flies that were returned to us to figure out which ones were truly the C-gene knockouts.
We knew this was going to be difficult, so we wanted to find a way to be able to see our knockdown flies without having to do lengthier molecular techniques, such as a polymerase chain reaction (PCR), on every fly. To this end we also constructed a plasmid with a sequence for ebony, a body-color marker. The expectation is that when one plasmid is successfully injected, the second one is as likely to be present--a co-CRISPR.
When we received the flies back from Best Gene, we mated them to a stock of flies we have in the lab. The children from those crosses should express the ebony body shade if they have a successful ebony mutation, and also hopefully the C gene mutation as well.
The next step that I'm doing now is to collect these guys and sequence their DNA to see if the C gene has been successfully altered.
Once we confirm we have flies that have gene C completely eliminated by CRISPR, we can keep a stock of these flies and later cross them with animals expressing both Toll and Alzheimer's A-beta. Will there be an even more dramatic rescue?
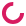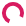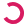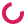
0 Comments Add a Comment?